 The results of bromination ( light-induced at 25 ✬ ) are even more surprising, with 2-bromopropane accounting for 97% of the mono-bromo product.ĬH 3-CH 2-CH 3 + Br 2 → 3% CH 3-CH 2-CH 2 Br + 97% CH 3-CH Br-CH 3 Light-induced gas phase chlorination at 25 ✬ gives 45% 1-chloropropane and 55% 2-chloropropane.ĬH 3-CH 2-CH 3 + Cl 2 → 45% CH 3-CH 2-CH 2 Cl + 55% CH 3-CH Cl-CH 3 If all these hydrogen atoms were equally reactive, halogenation should give a 3:1 ratio of 1-halopropane to 2-halopropane mono-halogenated products, reflecting the primary/secondary numbers. For example, propane has eight hydrogens, six of them being structurally equivalent primary, and the other two being secondary. All the hydrogens in a complex alkane do not exhibit equal reactivity. The halogenation of propane discloses an interesting feature of these reactions. 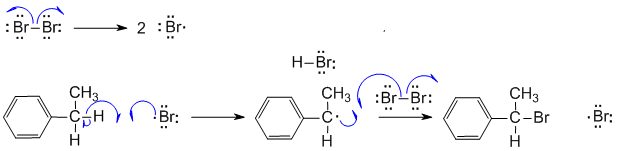
Can you write structural formulas for the four dichlorinated isomers? Four constitutionally isomeric dichlorinated products are possible, and five constitutional isomers exist for the trichlorinated propanes. Thus chlorination of propane gives both 1-chloropropane and 2-chloropropane as mono-chlorinated products. When alkanes larger than ethane are halogenated, isomeric products are formed. This is because hydrogen abstraction by bromine radical is much less exergonic than by chorine radical – and this in turn means that the transition state for abstraction by bromine resembles the resulting intermediate more closely than the transition state for abstraction by chlorine resembles its intermediate. 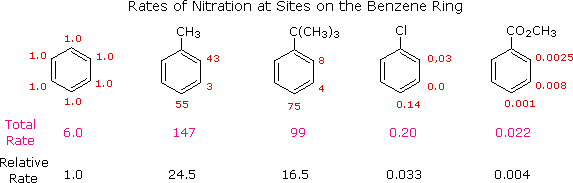
The regiochemical trends are the same as for chlorination, but significantly more pronounced (in other words, bromination is more regioselective). Unfortunately, chloroalkanes will readily undergo further chlorination resulting in polychlorinated products, so this is not generally a terribly useful reaction from a synthetic standpoint.Īlkanes can be brominated by a similar reaction. Recall that the Hammond postulate ( section 6.2, section 15.2B) tells us that a lower-energy intermediate implies a lower-energy transition state, and thus a faster reaction. This is because the tertiary radical intermediate is more stable than the secondary radical intermediate that results from abstraction of the proton on carbon #3, and of course both are more stable than a primary radical intermediate. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
